Aritrick Chatterjee1, Grace Lee2, Deb Dietz2, Aytekin Oto1, and Gregory Karczmar1
1Department of Radiology, University of Chicago, Chicago, IL, United States, 2Ingalls Memorial Hospital, Flossmoor, IL, United States
1Department of Radiology, University of Chicago, Chicago, IL, United States, 2Ingalls Memorial Hospital, Flossmoor, IL, United States
HM-MRI
was successfully implemented and validated to measure prostate tissue
composition on MRI scanners (Philips Achieva, Siemens Skyra) from different
vendors. Diagnostic accuracy for detecting PCa using HM-MRI was similar for
both MR vendors.
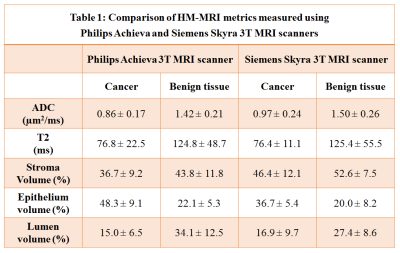
Table
1: Comparison of HM-MRI metrics measured using
Philips Achieva and Siemens Skyra
3T MRI scanners
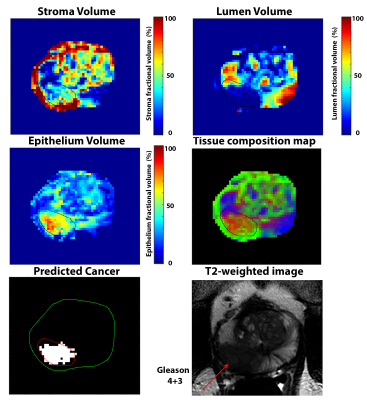
Figure
1: 56 years old patient with Gleason 4+3 cancer in right peripheral zone. The
cancer lesion (ROI) shows elevated epithelium volume (53.8%) and reduced lumen
(5.6%) and stroma (40.6%) volume and corresponding low ADC (0.618 um2/ms)
and T2 (87.5 ms) compared to surrounding benign tissue. HM-MRI correctly
predicts it be a clinically significant cancer on the predicted cancer map.
Images were taken on a 3T Philips Achieva scanner along with an endorectal
coil.
